
What are electric batteries and what types ?
Electric batteries definition and types, electric batteries are considered one of the basic components that are used in many different applications such as: cars, solar energy systems, and the operation of direct current loads.

Where the battery stores electrical energy with direct current to benefit from it in operating loads of electronic and electrical devices.
In this article, we will go into all the details about electric batteries, their definition and types.

Identification of the battery and its components:
A battery is a packet of chemical energy that can produce a limited amount of electrical energy when you need it, unlike regular electricity that runs through your home through wires coming from electric power stations. The battery converts the chemicals inside it into electrical energy, and it is used over days, weeks, months, or even years, depending on the type, size, and uses of the battery.
The basic unit of energy in a battery is called the cell, as batteries generally consist of one or more cells, and they consist of three sections:
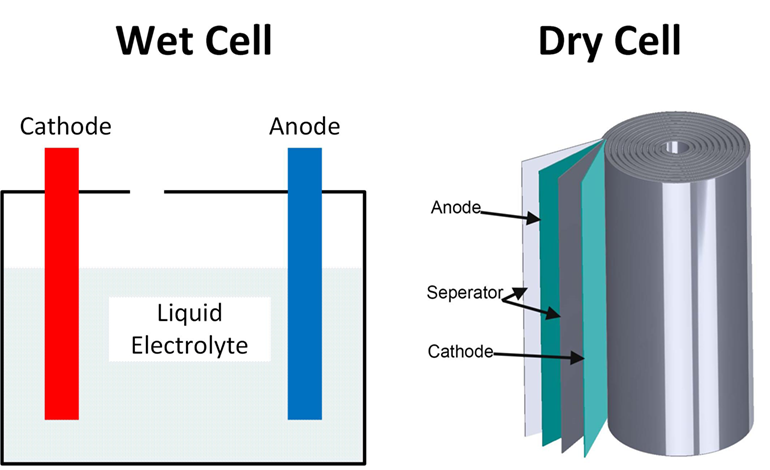
Anode: This is connected to the negative side of the battery, and is the terminal where electric current enters (or electrons leave the battery) during the discharge process.
Cathode: Connects to the positive terminal of the battery, which is the terminal from which electric current exits (or electrons enter the battery) during discharge.
Electrolyte: A liquid or gel-like chemical that separates the two electrodes (anode and cathode) and contains electrically charged particles.
Battery working principle:
It stores electric energy in the battery in the form of chemical energy during the charging phase, and converts the chemical energy inside the battery into electrical energy when connected to a load circuit, such as: 12-volt lighting LEDs.
The importance of electric batteries:
The importance of electric batteries lies in the storage of electrical energy in the form of direct current (DC) and not alternating current (AC), and electric batteries are widely used in cars, solar energy systems, and mobile devices of all kinds.
types of batteries
Batteries are classified into two categories:
A- Batteries that cannot be charged (primary):
Batteries are used for one time only and cannot be charged again.
This type of battery achieved a progress rate of approximately 23.6% of the global market until 2009 AD, but it began to decline due to the presence of secondary batteries that can be charged.

Primary battery uses:
Widely used in watches, remote controls, electronic switches, some lighting devices, military equipment…..etc.
Primary battery features:
B- Batteries that can be charged (secondary):
They are batteries that can be charged and discharged more than once.
This type of battery has made great progress in recent times and has become used in many fields and is indispensable.

Classification of secondary batteries:
Secondary batteries are classified according to chemicals into:
1- Lead acid batteries:
They are rechargeable batteries used at the present time, and they are the oldest types of batteries, as they have a high ability to provide electrical energy in different sizes and shapes and have a low cost, but not like lithium-ion batteries (Li-ion).
2- Lithium-ion batteries:
This type of battery is newer and has different chemicals than lead acid batteries. It is the preferred type for electronic devices and various loads because of its high energy density, long life, and high voltage, as its nominal voltage is 3.6V per cell.
3- Nickel Cadmium (Ni-Cd) Batteries:
It is another type of battery that can be recharged and discharged. It consists of positive and negative electrodes containing the chemicals of nickel oxide hydroxide and the metal cadmium. One of its advantages is the preservation of voltage and electric charge when not in use.
4- Nickel-metal hydride batteries (Ni-MH):
It is also a type of battery, which can be recharged and discharged for electrical energy. The chemical reaction at the positive electrode of the battery is similar to that of nickel-cadmium (Ni-Cd) cells, but the difference is at the negative electrode, where they use a hydrogen-absorbing alloy instead of nickel-cadmium (Ni-Cd).
Advantages of rechargeable batteries:
Disadvantages of rechargeable batteries:
In general, batteries play an important role in the field of electric energy for several reasons, namely:
Battery selection conditions:
Atlas Support and Services has distinguished practical experience in testing and testing services for electric batteries, starting from the stage of manufacturing electric batteries and ending with installation through a team of highly experienced engineers and technicians using the latest testing equipment and various tests. In addition, Atlas grants quality and safety certificates for various electric batteries and special international accreditation certificates With electric batteries such as ISO /IEC

We are happy to serve you and we are pleased to contact you to request our services or for any inquiries, send us an e-mail (info@atls.com.sa) or call us directly
(00966112080550)